This is international harmonized standard for Medical Devices that discusses about the possible risk factors, its latest version (ISO 14971:2019).
Keys for identifying, evaluating, and mitigating hazards incorporated with medical devices to demonstrate conformity to product safety and performance requirements. This article provides an overview of the standard, but should not be used as a substitute for the actual text of the standard.
As in the case of a quality management system, a risk management system addresses the full lifecycle of a medical device; including the design, manufacture, and use of the device. Also, while ISO 14971:2019 does not, itself, require the implementation of a quality management system, risk management is most often an important part of a strong quality management system.
Compliance ISO 14971:2019 requires that a risk management system be established and maintained throughout the product lifecycle, and that all processes and results are stored in a risk management file. The risk management system will include processes for risk analysis, evaluation, and control. It is important to note that the standard does not define acceptable levels of risk for medical devices – this is left to the manufacturer to determine as part of their risk management processes.
It is important for the organizational personnel to have proper understanding about product usage, process techniques including with expectation of desire results and particular harmonized standards for the evaluation of product life cycle to address specific risks and fully demonstration of the risk mitigation by covering the residues and warning need to inform to the users.
Risk evaluation
The latest version of the standard and guidance, however, emphasize that the matrix should be the output of the risk management policy, which would define the criteria for risk evaluation.
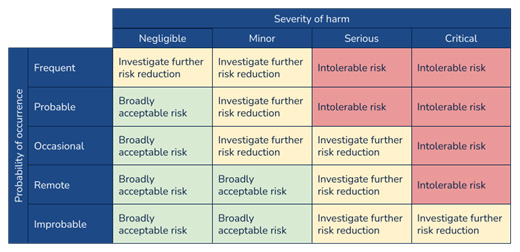
Risk Controls:
When a hazard is found to have an unacceptable risk level, risk control activities are put in place to mitigate the risk. ISO 14971:2019 requires that “state-of-the-art” best practices that are used for similar devices be employed. State-of-the-art does not necessarily mean the most advanced processes and technical features, but rather those that are generally accepted in the industry. Risk control options should include, in order of importance:
- Inherent safety by design and manufacture
- Protective measures built into the device or into the manufacturing process
- Provided safety information, and where appropriate, training to users
Risk/benefit analysis should be performed and where benefit is determined to outweigh risk, the manufacturer will need to decide what safety information is necessary to disclose.
Production and post-production information
A substantial change in ISO 14971:2019 standard is the expansion of requirements for production and post-production activities. The manufacturer will need to perform a full review of the risk management process prior to commercial distribution. The review should ensure that the risk management plan has been appropriately implemented, the overall risk is acceptable, and that procedures are in place to gather and maintain risk data during production and post-production of the medical device. ISO 14971:2019 aligns closely with the ISO 13485:2016 section 8 requirements for feedback, analysis of data and CAPA.
For further information or any other queries, please contact us.